Alkenes (Cambridge (CIE) IGCSE Co-ordinated Sciences (Double Award)): Revision Note
Exam code: 0654 & 0973
Did this video help you?
Catalytic cracking
Alkenes are unsaturated hydrocarbons
This means that they contain at least one carbon-carbon double bond (C=C)
The bonding in alkenes is covalent, as they consist only of non-metal atoms
Their general formula is CnH2n
Table of alkenes
Displayed formula | Name | Molecular formula |
|---|---|---|
 | ethene | C2H4 |
 | propene | C3H6 |
 | but-1-ene | C4H8 |
 | pent-1-ene | C5H10 |
The first four members of the alkene homologous series
Why alkenes are more reactive than alkanes
The carbon–carbon double bond can open up and form new bonds.
This allows alkenes to react more easily than alkanes, which only contain strong single covalent bonds.
Manufacture of alkenes and hydrogen (cracking)
Extended tier only
Although fractional distillation produces useful fractions, it produces:
Too many long chain hydrocarbons
Not enough short-chain hydrocarbons, such as petrol and alkenes
To solve this, long-chain alkane molecules are broken down into smaller, more useful molecules in a process called cracking
What is cracking?
Cracking is the thermal decomposition of long-chain alkanes into:
Shorter-chain alkanes
One or more alkenes
Hydrogen
Kerosene and diesel oil are often cracked to produce petrol, other alkenes and hydrogen
Conditions for cracking
Cracking requires:
A high temperature (around 600–700 °C)
A catalyst, usually aluminium oxide (alumina) or silicon dioxide (silica)
What happens during cracking
Long-chain alkane vapours are heated
The vapours pass over the hot catalyst
Carbon–carbon bonds break
A mixture of smaller alkanes, alkenes, and hydrogen is formed
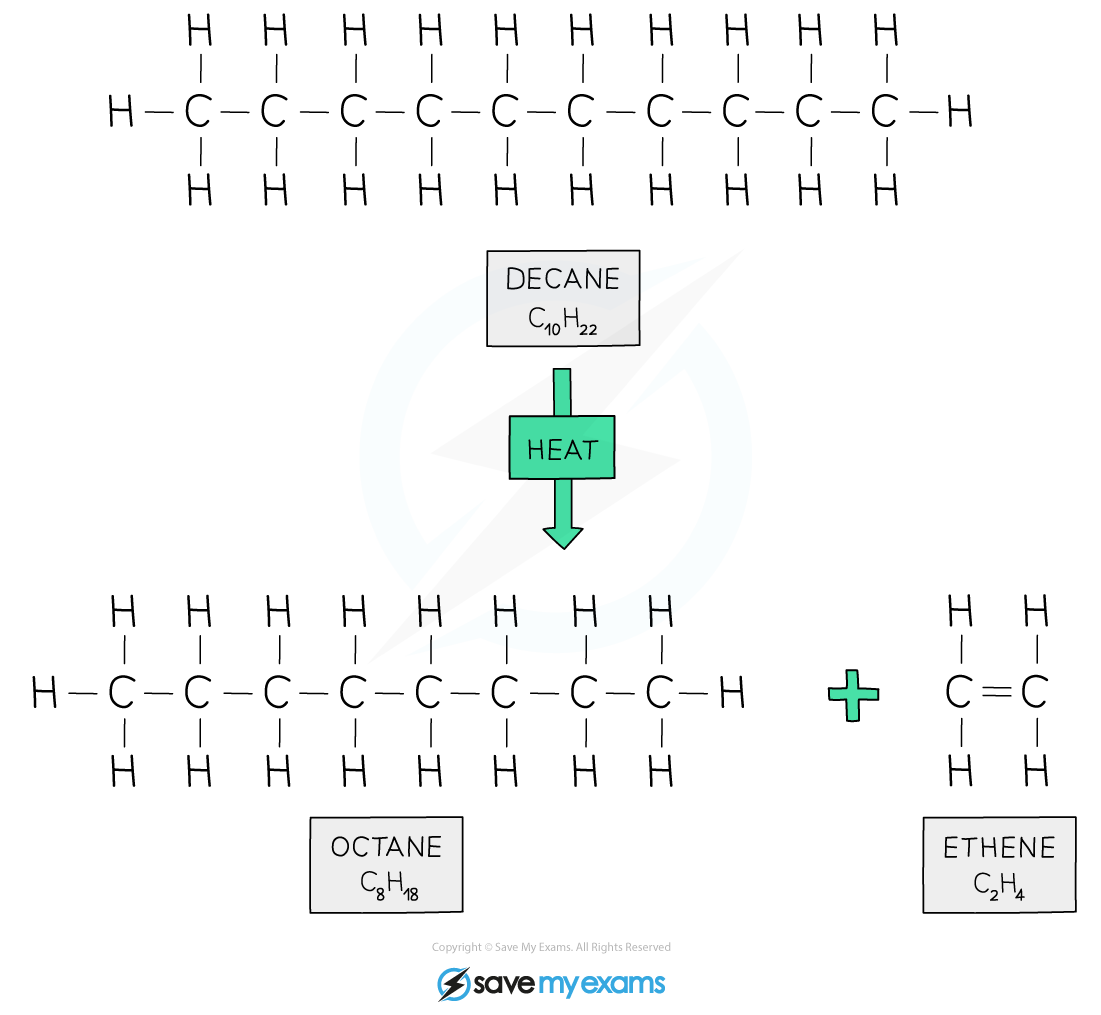
Example of cracking
A long-chain alkane can be cracked to form an alkene and a shorter alkane:
decane → octane + ethene
C10H22 → C8H18 + C2H4
Why cracking is important
Alkenes are used to make polymers
Hydrogen is used in processes such as ammonia manufacture
It increases the supply of useful fuels like petrol
Distinguishing from alkanes
The presence of a C=C double bond allows alkenes to be identified using aqueous bromine
Aqueous bromine, Br2 (aq), is an orange solution of bromine dissolved in water
It is often called bromine water
Testing with aqueous bromine
A few drops of aqueous bromine are added to the sample
The sample is then shaken
Results
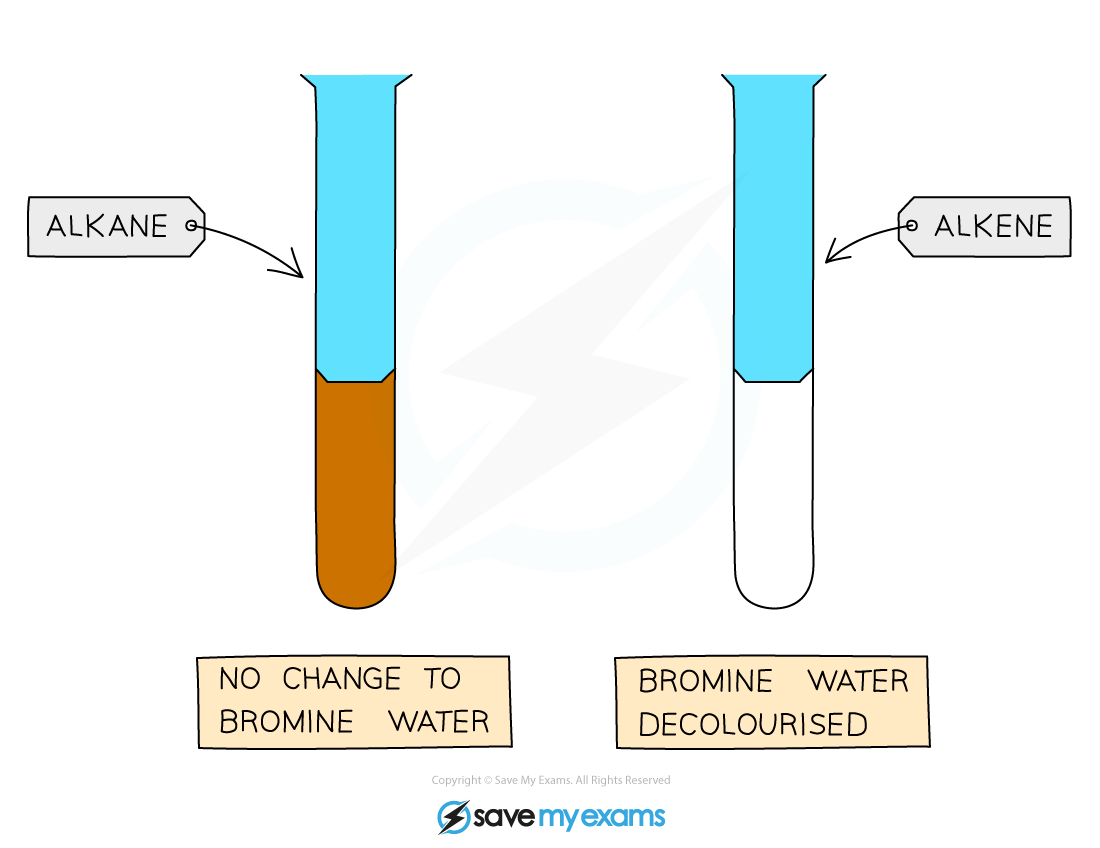
Alkane + aqueous bromine
No colour change
The solution remains orange
Alkene + aqueous bromine
Aqueous bromine is decolourised
Colour change: orange → colourless
This happens because bromine reacts with the C=C double bond and is no longer present in solution.
Examiner Tips and Tricks
Always say colourless, not clear, when describing the result with an alkene

Unlock more, it's free!
Was this revision note helpful?
