Pollution from Combustion (Edexcel A Level Chemistry) : Revision Note
Causes & Effects of Pollution
Alkanes are combusted (burnt) on a large scale for their use as fuels
Complete combustion
When alkanes are burnt in excess (plenty of) oxygen, complete combustion will take place and all carbon and hydrogen will be oxidised to carbon dioxide and water respectively
For example, the complete combustion of octane to carbon dioxide and water

The complete combustion of alkanes
Incomplete combustion
When alkanes are burnt in only a limited supply of oxygen, incomplete combustion will take place and not all the carbon is fully oxidised
Some carbon is only partially oxidised to form carbon monoxide
For example, the incomplete combustion of octane to form carbon monoxide

The incomplete combustion of alkanes
Incomplete combustion often takes place inside a car engine due to a limited amount of oxygen present
With a reduced supply of oxygen, carbon will be produced in the form of soot:

Car exhaust fumes include toxic gases such as carbon monoxide (CO), oxides of nitrogen (NO/NO2) and volatile organic compounds (VOCs)
When released into the atmosphere, these pollutants have serious environmental consequences damaging nature and health
Carbon monoxide
CO is a toxic and odourless gas which can cause dizziness, loss of consciousness and eventually death
The CO binds well to haemoglobin which therefore cannot bind oxygen and carbon dioxide
Oxygen is transported to organs
Carbon dioxide is removed as waste material from organs
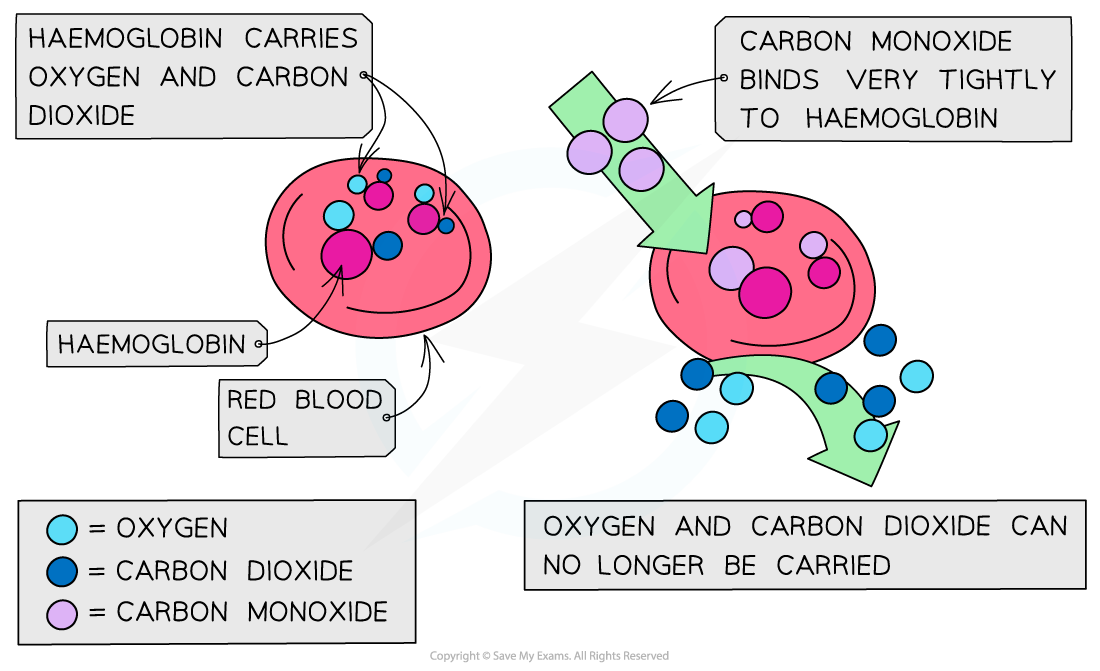
The high affinity of CO to haemoglobin prevents it from binding to O2 and CO2
Oxides of nitrogen
Normally, nitrogen is too unreactive to react with oxygen in air
However, in a car engine, high temperatures and pressures are reached causing the oxidation of nitrogen to take place:
N2(g) + O2(g) → 2NO(g)
N2(g) + 2O2(g) → 2NO2(g)
The oxides of nitrogen are then released in the exhaust fumes into the atmosphere
Car exhaust fumes also contain unburnt hydrocarbons from fuels and their oxides (VOCs)
In air, the nitrogen oxides can react with these VOCs to form peroxyacetyl nitrate (PAN) which is the main pollutant found in photochemical smog
PAN is also harmful to the lungs, eyes and plant-life
Nitrogen oxides can also dissolve and react in water with oxygen to form nitric acid which is a cause of acid rain
Acid rain can cause corrosion of buildings, endangers plant and aquatic life (as lakes and rivers become too acidic) as well as directly damaging human health
Particulates
Solid carbon particles (or particulates) released from incomplete combustion clump together to form soot which gradually falls back to the ground
Particulates can cause several problems:
If they are inhaled they can damage the lungs and cause respiratory problems
They can cover buildings and statues, making them look unclean and accelerating corrosion
They can reflect sunlight back into space reducing the amount of light reaching the earth, this is called global dimming
Pollutants, their Effect & Removal Table


You've read 0 of your 5 free revision notes this week
Sign up now. It’s free!
Did this page help you?
