Ester Hydrolysis (AQA A Level Chemistry): Revision Note
Exam code: 7405
Ester Hydrolysis
Hydrolysis of esters under acidic conditions
Hydrolysis is the reverse of the esterification reaction
Ester hydrolysis is an important reaction, for example, in the production of biodegradable plastics
Esters can be hydrolysed to reform a carboxylic acid and an alcohol (or the salt of a carboxylic acid) by heating with either a dilute acid, such as sulfuric acid, or an alkali, such as sodium hydroxide
When an ester is heated under reflux with dilute acid, an equilibrium mixture is formed
This means that the hydrolysis reaction does not go to completion.

Hydrolysis of esters under alkaline conditions
In contrast, heating an ester under reflux with a dilute alkali, such as sodium hydroxide, is an irreversible reaction.
The ester is fully hydrolysed, and the reaction goes to completion
The carboxylic acid initially formed reacts with the excess alkali to produce a carboxylate salt and an alcohol
To obtain the carboxylic acid, the sodium carboxylate salt must be acidified.
The carboxylate ion (–COO⁻) is protonated by an acid, such as hydrochloric acid (HCl), to form the carboxylic acid (–COOH)

Table showing differences in the hydrolysis of esters
Acid hydrolysis | Alkaline hydrolysis |
|---|---|
Equilibrium established/ does not go to completion | Reaction is irreversible/ goes to completion |
Heated under reflux with dilute acid (HCl or H2SO4) | Heated under reflux with dilute alkali (NaOH) |
Carboxylic acid and alcohol are produced | Carboxylate salt and alcohol are produced |
Worked Example
Name the products and write equations for the following hydrolysis reactions:
Ethyl ethanoate with a hot dilute sulfuric acid solution
Methyl propanoate with a hot sodium hydroxide solution
Answer:
Answer 1: Ethanoic acid and ethanol
CH3COOCH2CH3 + H2O ⇌ CH3COOH + CH3CH2OH
Answer 2: Sodium propanoate and methanol
CH3CH2COOCH3 + NaOH → CH3CH2COONa + CH3OH
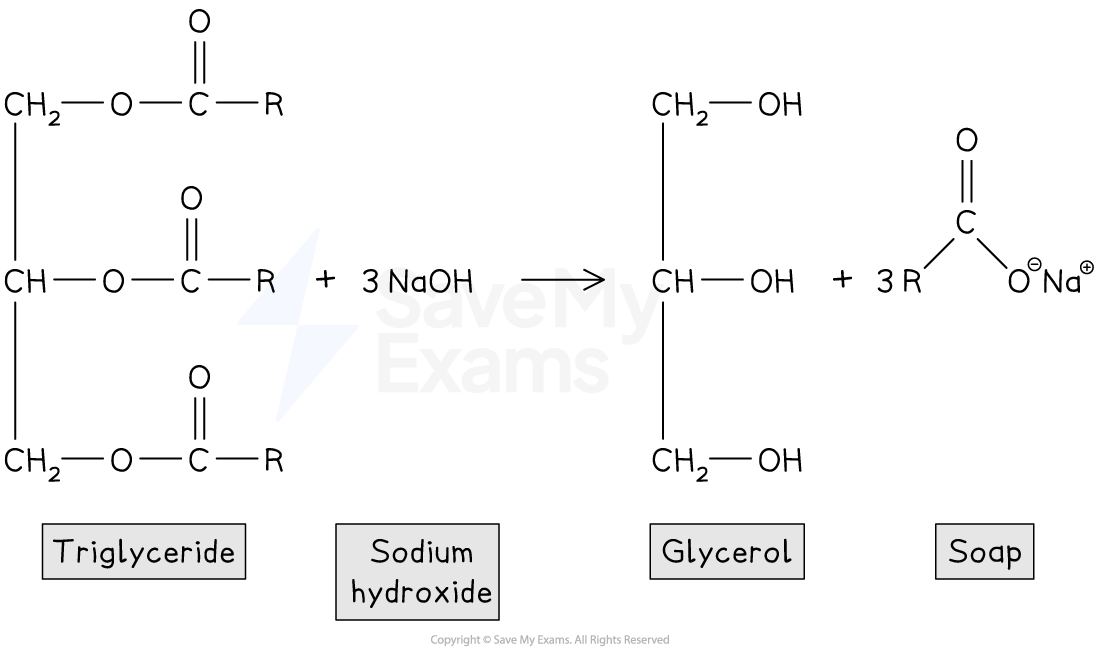
Making Soap
Soaps
Vegetable oils and animal fats can be hydrolysed in hot alkaline conditions using aqueous sodium hydroxide to form soaps
This process is known as saponification
Soaps are the sodium or potassium salts of long-chain carboxylic acids, which are known as fatty acids
When triglycerides (fats) are hydrolysed under hot alkaline conditions, the products are glycerol (propane-1,2,3-triol) and the salts of the fatty acids (soaps)

Unlock more, it's free!
Was this revision note helpful?
